












| Still deciding? Get samples of $ ! US$ 20/Piece Request Sample |
Medical Blister Packaging for Medical Instruments with CE ISO
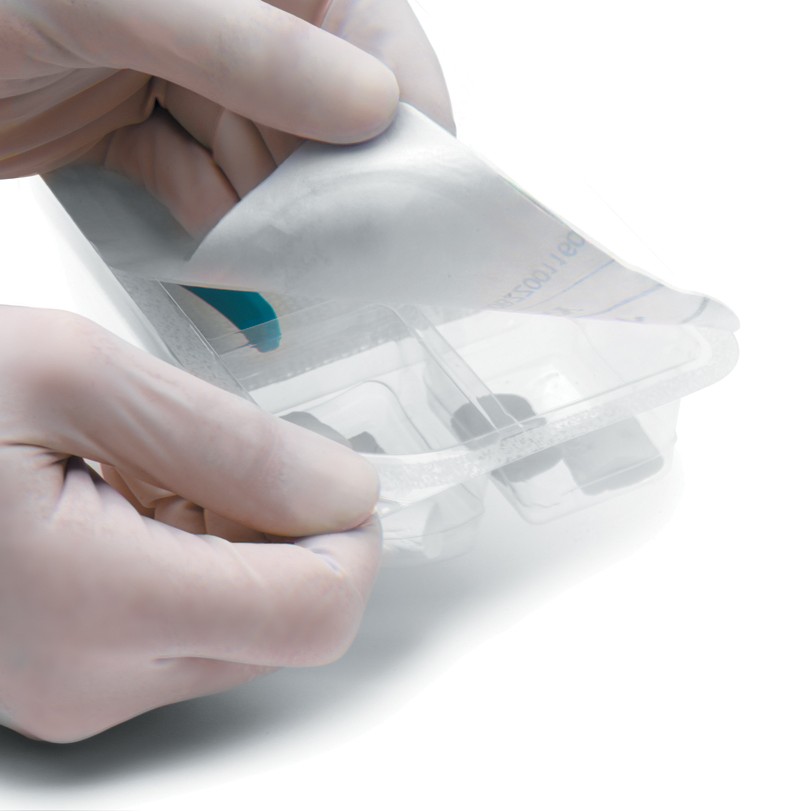
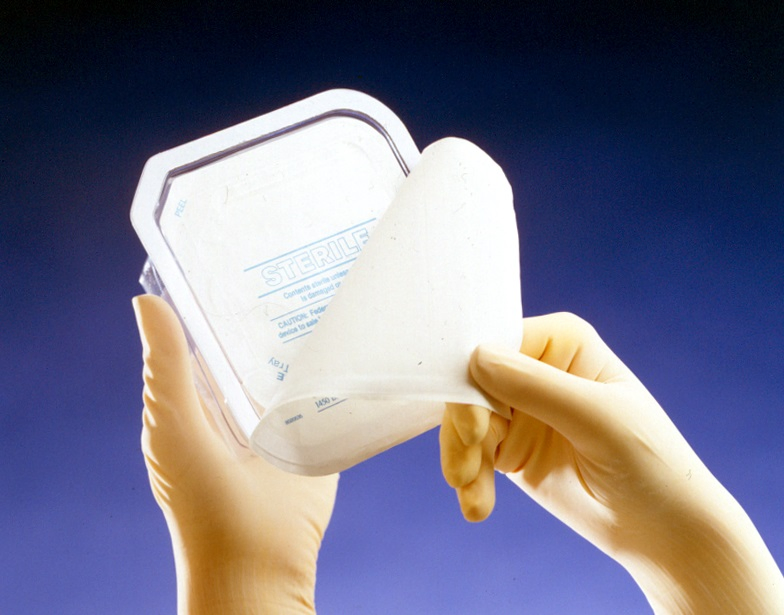
A cleanroom is an environment, typically used in surgical products packaging or scientific research, that has a low level of environmental pollutants such as dust, airborne microbes, aerosol particles and chemical vapors. More accurately, a cleanroom has a controlled level of contamination that is specified by the number of particles per cubic meter at a specified particle size.
To give perspective, the ambient air outside in a typical urban environment contains 35,000,000 particles per cubic meter in the size range 0.5 μM and larger in diameter, corresponding to an ISO 9 cleanroom, while an ISO 1 cleanroom allows no particles in that size range. The clean room is designed to be ISO certified as a Class 8/Class 100,000 clean room.





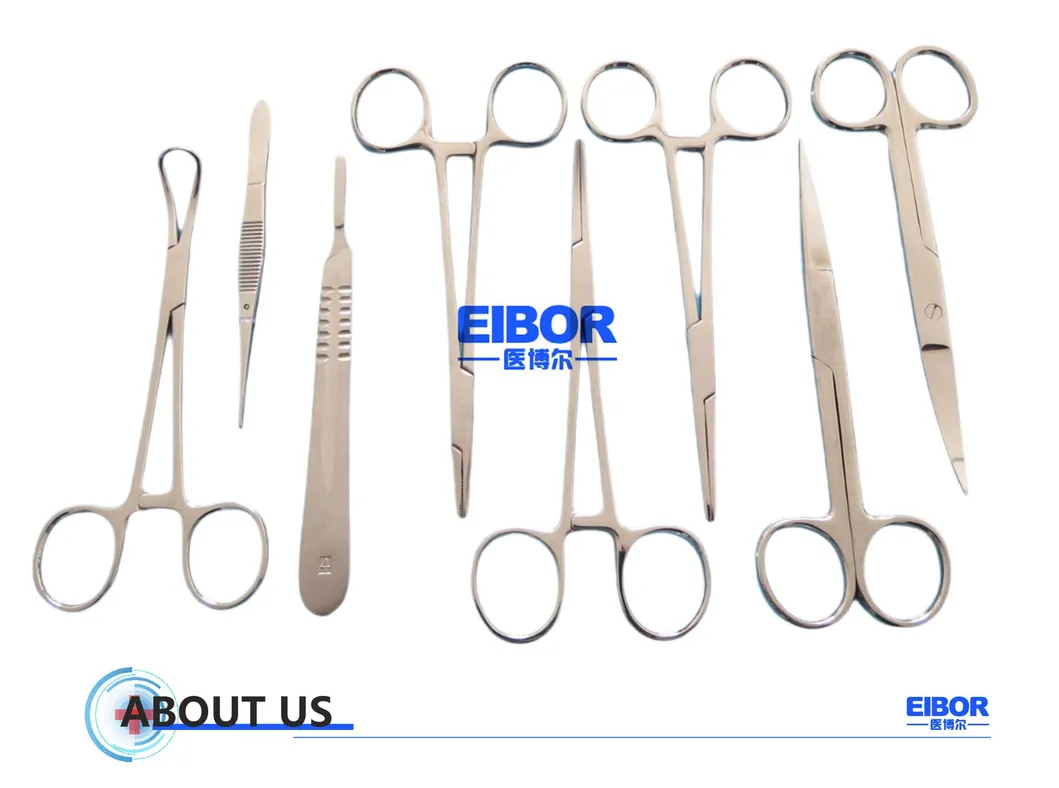
The manufacturer supplies various surgical instruments, both reusable and sterile disposable quality instruments, and is certified CE and ISO 13485:2016.
Medical packaging and sterilization services are provided, including ETO sterilization and Gamma radiation. The medical packing clean room reaches ISO 8, Grade 100,000 standards, ensuring that all valuable surgical instruments are packed in international standard medical clean rooms.
Strict QC management policies are followed, and all goods must be approved by the QC team before shipping. Quality, delivery time, and cost efficiency are the core focuses of production.



 Jalil Medical
Jalil Medical